Patient with Ebola-like symptoms alarms Canada as UK develops new vaccine
Published 10 Aug, 2014 03:09 | Updated 21 Sep, 2014 06:47
A vaccine against the Ebola virus has been developed by a British pharmaceutical company and is planned to be available by 2015, the WHO said. A patient with Ebola-like symptoms, who earlier sparked concern in Canada, has tested negative for the virus.
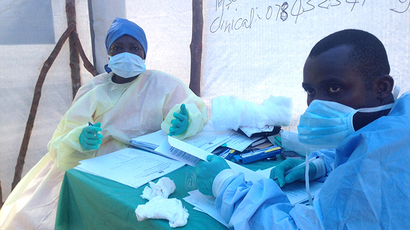
Ontario's Minister of Health and Long-Term Care Eric Hoskins confirmed that the man was “found to test negative for Ebola virus disease," is now doing well and it was anticipated he would enjoy a "full and speedy recovery."
The man has recently returned from a trip to Nigeria.
The male was hospitalized at William Osler Health System’s Brampton Civic Hospital, located in Toronto’s suburbs, on Friday.
“As a precautionary measure, Osler put in heightened infection control measures in the emergency department including isolating the patient,” the hospital said in a statement.
Doctors are “working closely” with public health officials “to confirm a diagnosis.”
“To date, there are no confirmed cases of Ebola in Ontario and the risk to Ontarians remains very low,” AFP quoted Ontario’s Interim Chief Medical Officer of Health, Graham Pollett, as saying.

Pollet assured that the province’s healthcare system “is prepared to respond should an individual arrive with symptoms that could suggest a disease, such as Ebola.”
He reminded that Ebola symptoms are similar to other common illnesses and added that doctors have been “advised to be on heightened alert for Ebola cases.”
Meanwhile, the World Health Organization (WHO) said on Saturday that a preventative vaccine against the Ebola virus has been developed by British pharmaceutical company GlaxoSmithKline, and could be available by 2015.
READ MORE: Intl health emergency: WHO issues Ebola warning
“We are targeting September for the start of clinical trials, first in the United States and certainly in African countries, since that’s where we have the cases,” AFP quoted WHO's head of vaccines and immunization, Jean-Marie Okwo Bele, as saying on French radio.
“And since this is an emergency, we can put emergency procedures in place...so that we can have a vaccine available by 2015,” he added.
At this point, there are several vaccines being tested. There is also a treatment called Zmapp, developed by San Diego-based Mapp Biopharmaceutical, which produced positive results on monkeys and may have helped two Americans recently infected in Africa.
READ MORE: ‘Secret serum’: Experimental Ebola drug used to treat 2 US aid workers
The current Ebola outbreak, which began in Guinea in March, is considered to be the world’s largest ever, with 961 deaths and nearly 1,800 people infected so far.
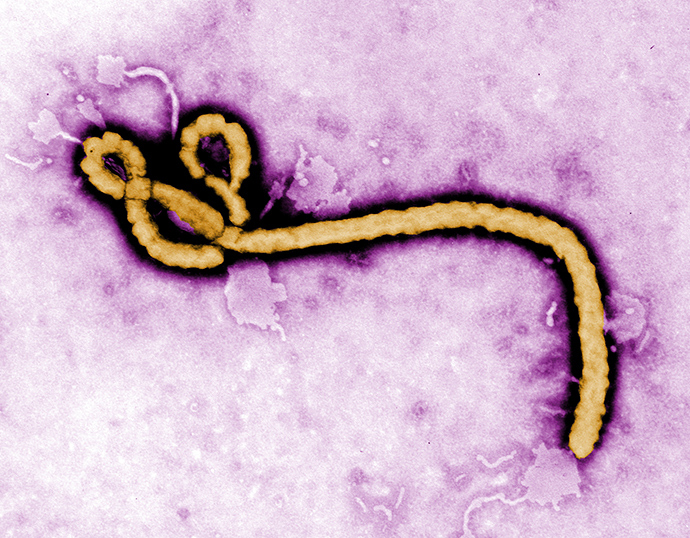
The worst hit countries so far include Sierra Leone, Liberia, and Guinea. Nigeria also reported nine confirmed cases of Ebola.
On Friday, Nigerian President Goodluck Jonathan declared a national emergency, following in the footsteps of Liberia’s president, who declared a state of emergency on Wednesday.
READ MORE: Liberia declares state of emergency as Ebola death toll rises to 932
The World Health Organization said on Friday that the Ebola outbreak in West Africa is an “extraordinary event” which poses a public health risk to other states. The health body described the consequences of a further international spread of the virus as “particularly serious” due to its virulence.
“A coordinated international response is deemed essential to stop and reverse the international spread of Ebola,” the WHO said in a statement after a two-day meeting of its emergency committee.